RFID ready for Lab 4.0
PRODUCTS > Laboratory

Sample Preparation
No more human erros.
Audit Trail
Seamless tracking of samples.
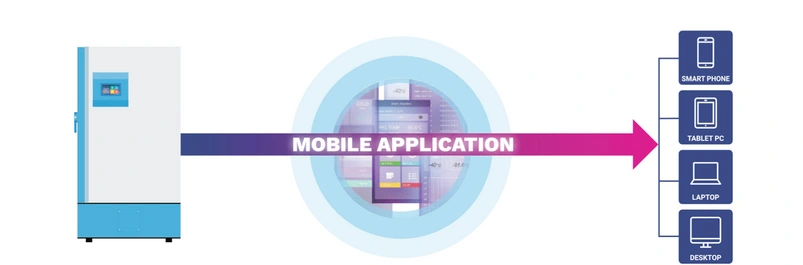
Laboratory 4.0
Sample controls the workflow.
OPTIMIZE PROCESSES
Radio Frequency Identification, RFID for short, represents a basic technology for Labor 4.0 applications. However, the fact that RFID is based on radio waves also makes it sensitive to external influences. It still requires fine-tuning of antennas and readers to achieve a high success rate in daily use. With proper planning, however, the power of RFID technology goes far beyond that of a mere barcode replacement. The bad news is that there is no one-size-fits-all solution for RFID, and there may never be. Implementing the right technology and developing the right software to apply RFID in the lab is our specialty. More details can be found on our XTC-ID project page.
Today's challenge - finding the right solution
Each customer's RFID requirements are specific and usually unique. We are specialized in adapting the available RFID technologies and tailoring a solution to your needs. We offer a complete package, from hardware to software solution , including project planning, analysis and implementation support to ensure that the integration of automatic identification (Auto ID) as well as digital data capture into existing processes runs smoothly.
For the medical and pharmaceutical sector, our patented xtID technology enables the inventory of vaccines, active ingredients, sample material and the like, at low temperatures down to -196°C, without interrupting the cold chain. Likewise, the retrofitting of commercial freezers (Ultra Low Freezer - ULF), with an automatic RFID inventory at temperatures down to -85°C is possible. More details about such low temperature applications can be found on the website of our new company xtID B.V..
AUTOMATE PROCESSES
The large storage capacity of modern RFID TAGs, in addition to obilgatory sample data, also allows the storage of method parameters, time stamps and access trials. This allows automated sample processing that almost completely eliminates human error sources and enables a fully digital track & trace according to FDA Standard CFR 21/11. The fact that important data is physically located on the sample itself also ensures protection against falsification or mix-ups, which means that management of reserve samples, for example, can be significantly optimized.
Our hardware components are specially designed for use in laboratories. The XTC-ID reader systems have an antenna multiplexer that enables different tray formats with capacities of up to 25 vials per module. The external connection of several antennas is also provided for example, to equip peripheral devices with RFID as well. The integration of our RFID technology into the PAL platform, including Chronos software control, allows easy integration and upgrade into existing workflows.